The Biden administration has been quietly telling states to prepare to vaccinate younger children against COVID-19 next month.
Recently, Pfizer-BioNTech submitted an application the U.S. Food and Drug Administration (FDA) to expand emergency use of its Covid vaccine to include kids between ages five and 11.
An unnamed White House official told NBC News that the federal government ahs been telling state governors that it anticipates the authorization will come in the next few weeks, as soon as early November.
What’s more, an official in the Department of Health and Human Services told the news outlet that Biden administration has already purchased 65 million pediatric doses of Pfizer’s vaccine.
That’s more than the two doses needed to fully vaccinate the 29 million children in the U.S. who would now become eligible.

The White House has been contacting state governors to tell them to prepare to start vaccinating children against COVID-19. Pictured: Marisol Gerardo, 9, is held by her mother as she gets a shot in the Pfizer Covid trial at Duke Health in Durham, North Carolina, April 2021

The Biden administration believes an FDA decision on Pfizer-BioNTech’s Covid vaccine in kids could come as soon as early November. Pictured: Biden speaks after signing three proclamations restoring protections for national monuments, October 2021
According to clinicaltrials.gov, Pfizer’s study in younger children worked similarly to the way it did in older children and adults.
A total of 4,500 younger kids aged six months and older were enrolled at nearly 100 clinical trial sites in 26 U.S. states, Finland, Poland and Spain.
Of those children, 2,268 were between ages five and 11.
About half of those in the five-to-11 group were given two doses 21 days apart and the other half were given placebo shots.
The team then tested the safety, tolerability and immune response generated by the vaccine by measuring antibody levels in the young subjects.
Pfizer said it had selected lower doses for COVID-19 vaccine trials in children than are given to teenagers and adults.
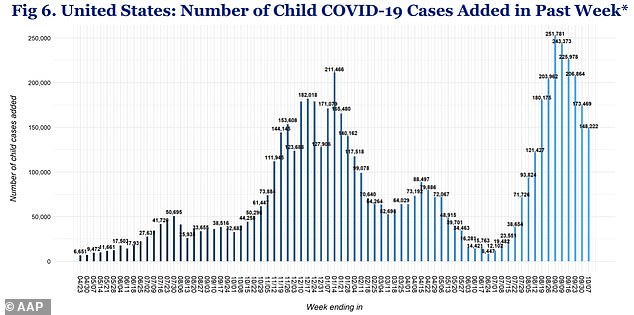
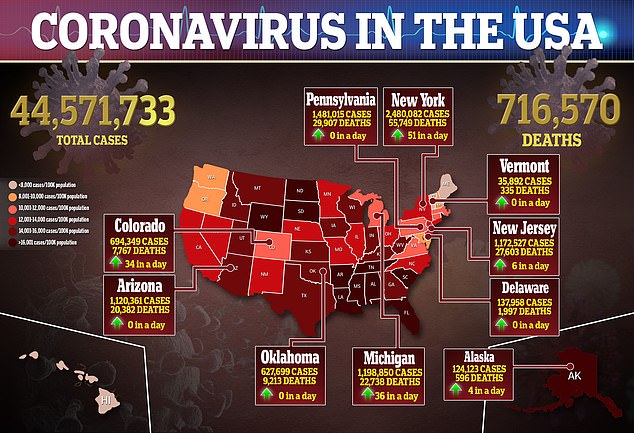
Weekly child Covid cases reached more than 243,000 in early September, but have since fallen to 148,000 as of last week *above)
Those aged 12 and older receive two 30 microgram (μg) doses of the vaccine.
However, children between ages five and 11 were given 10 μg doses and kids from six months to four years old received three μg doses.
Unlike the larger clinical trial conducted in adults, the pediatric trial did not measure efficacy by comparing the number of COVID-19 cases among the vaccine group to the number in the placebo group.
Instead, scientists looked at levels of neutralizing antibodies in young vaccine recipients and compared the levels to those seen in adults.
The companies expect data on how well the vaccine works in children between ages two and five and between six months and two years of age by the end of the year.
Recently, pediatric cases increased from 71,000 per week at the beginning of August to more than 243,000 in early September, fueled by the Delta variant.

However, they now appear to be trending downward with about 148,000 reported last week, according to the American Academy of Pediatrics.
There have also been 520 pediatric deaths since the start of the pandemic, indicating children make up less than 0.1 percent of all deaths.
Currently, no evidence suggests the Delta variant is more dangerous in kids than previous strains of the virus.
Because of this low risk of severe illness, polls have shown that many parents are not inclined to vaccinate their children.
A July 2021 survey, conducted by CS Mott Children’s Hospital National Poll on Children’s Health at Michigan Medicine last month, found that 39 percent of parents said their children already gotten a coronavirus shot.
However, 40 percent of parents also said it was ‘unlikely’ that their children would be getting vaccinated.’
Another poll from Axios/Ipsos in September found that 44 percent of parents of children aged five to 11 said their kids were likely to get a vaccine and 42 percent said it was unlikely their children would be immunized.
Source link : https://www.dailymail.co.uk/health/article-10089257/Biden-administration-quietly-telling-states-prepare-vaccinate-kids-soon-early-November.html